
DNA, RNA, proteins… All of these macromolecules can be analyzed using the electrokinetic process electrophoresis. In a 2020 webinar hosted by the American Institute of Chemical Engineers (AIChE), Professor Cornelius Ivory from Washington State University went over the four modes of electrophoresis: zone electrophoresis, moving-boundary electrophoresis, isotachophoresis, and isoelectric focusing — and discussed modeling them in the COMSOL Multiphysics® software.
Electrophoresis Overview
Electrophoresis is a general term used to describe the movement of charged particles in a fluid under the influence of an electric field. The word itself is derived from Greek; electro referring to electricity and phoresis meaning “to carry” or “to bear”.
One of the first observations of the electrokinetic process occurred over 200 years ago, in 1801, by French chemist Nicolas Gautherot. At the time, Gautherot saw electrophoresis taking place in a water droplet that was placed between two electrically attached metal plates (called electrodes).
Today, electrophoresis is commonly used to separate molecules based on their charge and size. This method can accurately separate macromolecules, such as DNA, RNA, proteins, nucleic acids, and plasmids.
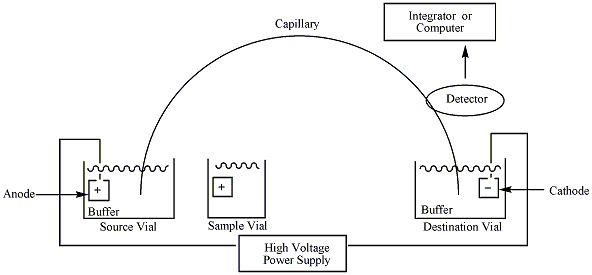
Sketch of capillary electrophoresis (CE). Image by Apblum. Licensed under CC BY-SA 3.0, via Wikimedia Commons.
The diagram above shows capillary electrophoresis (CE) — a modern technique used to detect short tandem repeat alleles in forensic DNA laboratories.
To get a comprehensive look at the mathematics behind electrophoresis, a good resource to start with is The Dynamics of Electrophoresis by Richard Mosher, Dudley Saville, and Wolfgang Thormann.
The 4 Basic Modes of Electrophoresis
The four basic modes of electrophoresis are:
- Zone electrophoresis
- Moving-boundary electrophoresis
- Isotachophoresis
- Isoelectric focusing
In most cases, the physics of new electrophoretic methods can be related back to these four modes.
Zone Electrophoresis
Zone electrophoresis is often used by scientists to analyze biopolymers, nucleic acids, and proteins. This type of electrophoresis became widely used in the 1960s, thanks to the discovery of discontinuous buffers, and it is still the most popular mode in use today.
Moving-Boundary Electrophoresis
Moving-boundary electrophoresis was developed in the 1930s by Swedish biochemist Arne Tiselius, who eventually went on to earn the Nobel Prize in Chemistry in 1948 for his work on electrophoresis and absorption analysis. This method is particularly beneficial when analyzing larger molecules, like proteins, which can be difficult to separate due to their differing charges. Historically, this method played an important role in moving the study of electrophoresis forward, but it isn’t widely applied today.
Isotachophoresis
Isotachophoresis is based on moving-boundary electrophoresis, and it is a very powerful separation method. This technique has been around since the 1920s and is still widely applied in research today. For example, isotachophoresis is used in the analysis of organic acids in silage, anions in urine and serum, inorganic ions in water, proteins, and amino acids.
Isoelectric Focusing
The fourth and final mode discussed in the AIChE webinar by Professor Ivory, isoelectric focusing, is often used in biological separations. In a previous blog post, we discussed how this type of electrophoresis is especially valued for its ability to concentrate and focus molecules into a fixed state.
How are these four modes the same and how are they different?
Modeling Electrophoresis Using COMSOL Multiphysics®
In the AIChE webinar, Professor Ivory discussed how the bulk equations for modeling all modes of electrophoresis are always the same, but the initial conditions and boundary conditions vary from type to type. Ivory turned to COMSOL Multiphysics to model the four basic modes.
Each model contains a cathodic reservoir, an anodic reservoir, and a channel; the cathodic and anodic reservoirs are described as boundary conditions, while the channel is the modeling domain. The initial conditions differ depending on which mode is used, and the models are all time-dependent, with the independent variables being the position in the channel and time.
In the table below, you can find the cathodic reservoir, anodic reservoir, and channel used for each mode. The governing equations in the channels are the Nernst–Planck equations in combination with the electroneutrality condition, for all four modes.
| Mode | Cathodic Reservoir | Anodic Reservoir | Channel |
|---|---|---|---|
| Zone electrophoresis | Running buffer | Running buffer | Running buffer and sample (which should be dialyzed in practice) |
| Moving-boundary electrophoresis | Sample | Leading electrolyte | Leading electrolyte |
| Isotachophoresis | Trailing electrolyte | Leading electrolyte | Trailing electrolyte, sample, and leading electrolyte |
| Isoelectric focusing | Basic solution (high pH) | Acidic solution (low pH) | Carrier ampholytes and sample |
For each model, Ivory accounts for seven dependent variables (ten for isoelectric focusing), including six chemical species (nine for isoelectric focusing) and the electric potential in the channel:
- Sodium (Na)
- Chlorine (Cl)
- Tris (Tr)
- Acitate (Ac)
- Seperand 1 (Sp1)
- Seperand 2 (Sp2)
- Electric potential
- Two pseudo-ampholytes and tricine for isoelectric focusing
The different solutions’ pH in the channel is predefined, as a constant value or as a given pH profile (isoelectric focusing). In addition, each model requires the following input parameters and initial conditions:
- Diffusion coefficients
- Mobilities
- Acid-base relationships
- Reservoir concentrations
- Operating voltages
- Initial channel concentration profiles
- Initial electric potential profile
Below, you can see each mode simulated in COMSOL Multiphysics. The plots show the concentration and pH in the channel at different given times after the sample enters the channel. We can here see the qualitatively different shapes of the sample profiles (red and blue).
Clockwise from top left: zone electrophoresis, moving-boundary electrophoresis, isoelectric focusing, and isotachophoresis.
Try It Yourself
Professor Ivory created 2 downloadable MPH files for the electrophoresis models discussed above; one of the files focuses on zone electrophoresis modeling and the other contains complete models for all four basic modes of electrophoresis.
You can find and download the files on the Application Exchange via the button below.






Comments (0)